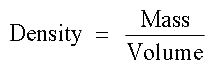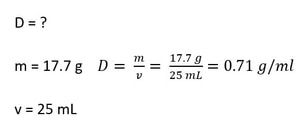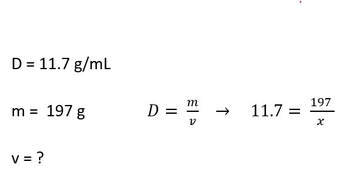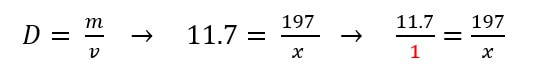Density
A physical property is a property of matter that can be observed without changing the make up of the substance. The material may change state (to solid, liquid or gas), but the material retains the same chemical make-up. These properties are essentially anything you can tell by observing it with your five senses.
Density
One of the most commonly used physical properties for chemistry is density. Density tells us how close the particles are to one another in a substance. The closer the particles are to one another, the greater the density.
Density
One of the most commonly used physical properties for chemistry is density. Density tells us how close the particles are to one another in a substance. The closer the particles are to one another, the greater the density.
Density is calculated using the following equation:
Typical units for density are g/ml and g/cm3, although it is not uncommon to see densities expressed in g/L for gases.
The density of water is 1.0 g/ml.
An object with a density greater than the object it is in will sink. An object with a density less than the object it is in will float. For example, the denisty of oil is less than that of water, so when you pour the two liquids together, layers will form, with the oil on top and the water on the bottom. The oil in this case “floats” and the water “sinks”. Lead, however, has a density greater than 1.0 g/ml, so when lead is put in water, the lead will “sink” to the bottom, while the water will “float”.
When using the equation, you may have to find any one of the three items within the formula: Density, mass or volume. Here are a couple of examples:
1) What is the density of a liquid that has a mass of 17.7 g when 25 mL of the liquid is placed in a graduated cylinder?
An object with a density greater than the object it is in will sink. An object with a density less than the object it is in will float. For example, the denisty of oil is less than that of water, so when you pour the two liquids together, layers will form, with the oil on top and the water on the bottom. The oil in this case “floats” and the water “sinks”. Lead, however, has a density greater than 1.0 g/ml, so when lead is put in water, the lead will “sink” to the bottom, while the water will “float”.
When using the equation, you may have to find any one of the three items within the formula: Density, mass or volume. Here are a couple of examples:
1) What is the density of a liquid that has a mass of 17.7 g when 25 mL of the liquid is placed in a graduated cylinder?
Lead has a density of 11.7 g/mL. How much volume will a piece of lead have when its mass is 197 g?
This is a hard problem to solve, but it you put the density over 1, you have two fractions that you can cross-multiply.
Then you can cross-multiply and divide.