Types of Elements
On the periodic table, there are three types of elements: metals, nonmetals and metalloids. These types are categorized by their properties.
Metals have the following properties:
Nonmetals have the following properties:
Metalloids have properties of both metals and nonmetals. These elements are a buffer between the metals and nonmetals. These elements are defined as having some properties of both metals and nonmetals. A metalloid may be shiny, but not conduct electricity and break when hit with a hammer. Or a metalloid may be malleable and shiny, but not react with acids.
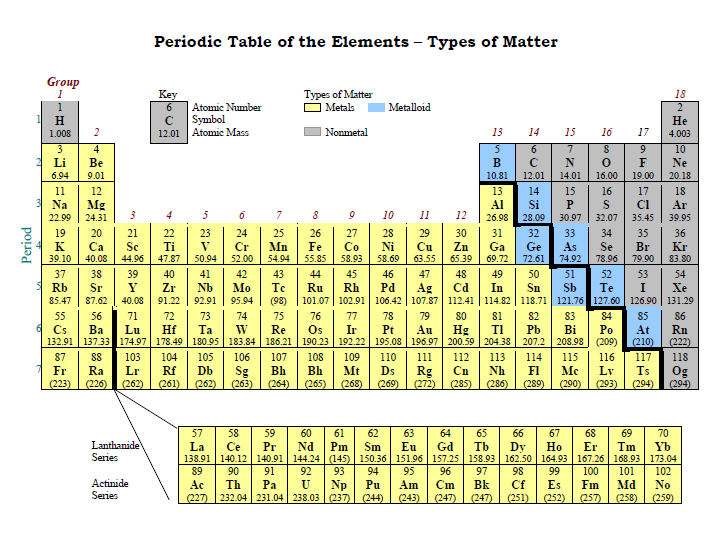
The following periodic table divides the elements of the periodic table into their three types.
Metals have the following properties:
- metals have a luster or shine
- metals are good conductors of heat/electricity
- most metals are solids at room temperature (all except mercury)
- metals are malleable, or can be shaped or bent and the metal will not break.
- most metals will react with acids
Nonmetals have the following properties:
- nonmetals do not have a luster; they are dull in appearance
- nonmetals are poor conductors of heat and electricity
- nonmetals are not malleable: when pressure is placed on them, the nonmetals will break.
- nonmetals can be solid, liquid or gas at room temperature
- most nonmetals will not react with acids
Metalloids have properties of both metals and nonmetals. These elements are a buffer between the metals and nonmetals. These elements are defined as having some properties of both metals and nonmetals. A metalloid may be shiny, but not conduct electricity and break when hit with a hammer. Or a metalloid may be malleable and shiny, but not react with acids.
The following periodic table divides the elements of the periodic table into their three types.