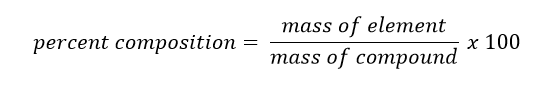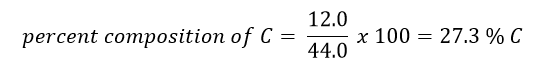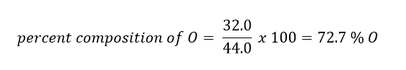Percent Composition
Percent Composition
It is often necessary to know how much of a particular element is in a compound. The percent composition of the mass made up by each element in a compound is called the percent composition of that element. Percent composition can be calculated using the following formula:
It is often necessary to know how much of a particular element is in a compound. The percent composition of the mass made up by each element in a compound is called the percent composition of that element. Percent composition can be calculated using the following formula:
Example: Suppose you had a mole of CO2. What is the percent composition of each element?
Answer: In one mole of CO2, there is 12.0 g of C (due to the one carbon in the compound) and 32.0 g of O (due to two oxygens in the compound - 2 x 16.0 g = 32.0 g). The total mass of one mole of CO2 is therefore 44.0 g (12.0 g C + 32.0 g O).
Answer: In one mole of CO2, there is 12.0 g of C (due to the one carbon in the compound) and 32.0 g of O (due to two oxygens in the compound - 2 x 16.0 g = 32.0 g). The total mass of one mole of CO2 is therefore 44.0 g (12.0 g C + 32.0 g O).