ACID Rain
Acid rain and precipitation
Acid precipitation is rain, fog or snow) with a pH in the acidic range of the pH scale (typically with a pH below 5.6).
Acid precipitation is rain, fog or snow) with a pH in the acidic range of the pH scale (typically with a pH below 5.6).
|
Acid precipitation is caused by the reaction of SO2, NOx, and CO2 with water in the atmosphere:
Examples: SO2 (g) + H2O (g) --> H2SO3 (aq)
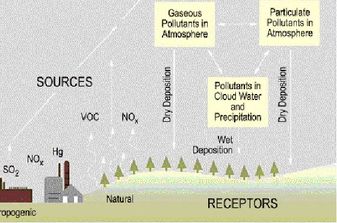
CO2 (g) + H2O (g) --> H2CO3 (aq) NOx (g) + H2O (g) --> HNOx+1 (aq) SO2 and NOx come from the combustion of fossil fuels or industrial process, while CO2 is mostly natural, but is increased by human activities. See diagram to see some of the sources of acid pollution:
Effects of acid precipitation: 1) Destruction of trees and plants sensitive to changes in pH. 2) Alteration of the pH of certain bodies of water, 3) Death of water animals sensitive to low pH. 4) Sores on skin due to overexposure to acids. |