Properties of Water
|
Water is one of the rarest and most unusual substances in the universe. The abundance of water on Earth’s surface is unmatched by any planet or moon in our solar system. 70% of Earth’s surface is made up of water. Water is very important to the lives of plants and animals on this planet. Therefore, it is important to look at the properties of water and what it can do.
Property #1: The density of liquid water is 1.00 g/mL. Originally, the idea of density was based off of water, so these units of grams and milliliters is somewhat based on that. The density of solid water (ice) is 0.917 g/mL. The fact that solid water's density is less than the liquid version is fairly unique. Most other substances have a solid density more than the liquid. |
|
Property #2: Water freezes at 0 OC and boils at 100 OC. These numbers may seem perfect, and that is actually the design. The Celsius scale was developed based off of water, so that's why the numbers are so perfect.
Property #3: Water is often called the universal solvent. It can dissolve a lot of substances. Salt, sugar, baking soda, carbon dioxide - substances with very different physical and chemical properties can all dissolve in water. But water cannot dissolve all things. Oil, sand, some rocks, nitrogen do not dissolve very much or at all in water. Property #4: Water has electrical properties. When you have plain water, it will conduct a very small amount of electricity. But, when you dissolve an ionic compound in water, it will conduct quite a bit of electricity. This is because water is a polar substance. |
|
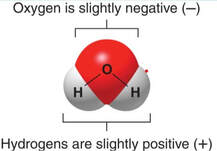
Property #5: Water is a polar molecule. In water, the hydrogen and oxygen share electrons in covalent bonds. However, the electron sharing to achieve and octet is not equal, and the electrons are more drawn to the O instead of the Hs. As a result, the O end of the molecule is slightly negative and the H end is slightly positive. This causes the water to be polar, which leads to other properties.
Property #6: Liquid and solid water is held together by hydrogen bonding. Hydrogen bonding is the strong intermolecular force that occurs when the positive end of one water molecule is attracted to the negative end of a neighboring water molecule in a liquid. This attraction causes the water molecules to be attracted and held together very strongly in a solid and liquid. Property #7: Water exhibits cohesion, which causes surface tension and high boiling points. Water's strong hydrogen bonding means that water particles are held tightly together in a liquid. This tight attraction has a term called cohesion. This cohesion causes surface tension, a tight film at the surface where molecules are help more tightly than below. Surface tension causes water to bead and will allow small objects to sit on the surface of water. |