Solutions and Concentrations
Solutions
A solution is a homogeneous mixture of two or more substances in a single physical state. There are several properties of solutions:
1) Particles in solution are very small, usually atoms, molecules or ions.
2) Particles are evenly distributed. Every sample of the solution will have the same concentration or ratio of particles as every other sample of the same solution.
3) Particles will not separate out in a solution. Everything stays mixed up evenly.
There are two parts to a solution:
1) Solute - the substance that dissolves. It becomes similar to the solvent. In Kool-Aid, the powder and the sugar are the solutes.
2) Solvent - the substance the solute is dissolving into. In Kool-Aid, the water is the solvent.
There are 4 types of solutions:
1) Solid solutions, also called alloys. Examples are a gold ring, steel, brass.
2) Gas solutions. Example is air
3) Liquid solutions - Examples are gasoline, motor oil.
4) Aqueous solutions, where water is the solvent. These include, orange juice, milk, ocean water.
A solution is a homogeneous mixture of two or more substances in a single physical state. There are several properties of solutions:
1) Particles in solution are very small, usually atoms, molecules or ions.
2) Particles are evenly distributed. Every sample of the solution will have the same concentration or ratio of particles as every other sample of the same solution.
3) Particles will not separate out in a solution. Everything stays mixed up evenly.
There are two parts to a solution:
1) Solute - the substance that dissolves. It becomes similar to the solvent. In Kool-Aid, the powder and the sugar are the solutes.
2) Solvent - the substance the solute is dissolving into. In Kool-Aid, the water is the solvent.
There are 4 types of solutions:
1) Solid solutions, also called alloys. Examples are a gold ring, steel, brass.
2) Gas solutions. Example is air
3) Liquid solutions - Examples are gasoline, motor oil.
4) Aqueous solutions, where water is the solvent. These include, orange juice, milk, ocean water.
The Process of Dissolving
When a solute dissolves in a solvent, it looks like it disappears. If the original solute is colored, it may change the color of the solvent. But most white ionic solids will remain colorless when dissolved in a clear liquid, like water. So, how do we know it is still there. Well, for something like salt or sugar in water, we can taste the solution and tell that the solute is still present because it tastes salty (or sweet for sugar). So, what has happened to the solute. It has disappeared, but we know it is still there.
When a solute dissolves in a solvent, it looks like it disappears. If the original solute is colored, it may change the color of the solvent. But most white ionic solids will remain colorless when dissolved in a clear liquid, like water. So, how do we know it is still there. Well, for something like salt or sugar in water, we can taste the solution and tell that the solute is still present because it tastes salty (or sweet for sugar). So, what has happened to the solute. It has disappeared, but we know it is still there.
|
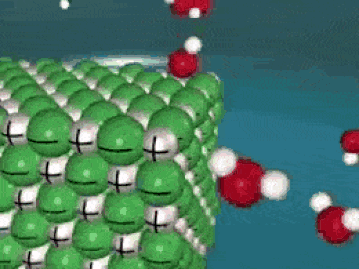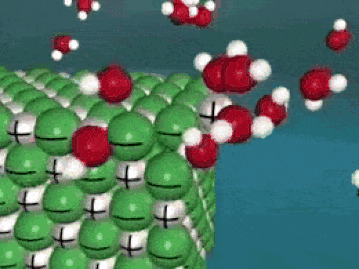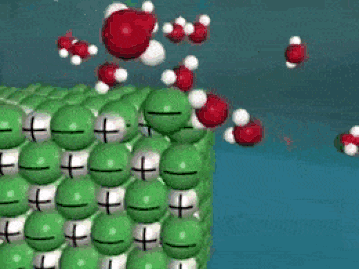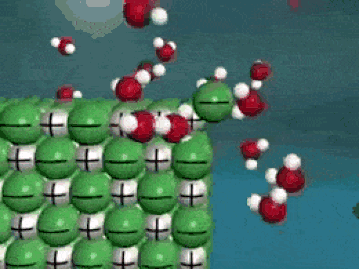
Dissolving is a process where the solute is taken apart into such small parts that they cannot be seen. For a molecular substance (held together by covalent bonds), the solute is down to the molecule size. For ionic compounds, the solute (in water) has been broken up into ions (even smaller than molecules). This process is a very violent process on the molecular level, as the solute is essentially ripped apart into smaller and smaller pieces until they cannot be seen.
As seen in the gif, water molecules come into the crystal and start taking the solute apart, piece by piece. The negative O of the water molecule grab the positives, and the positive H of the water molecules grab the negatives. This keeps happening over and over until the salt crystal is completely torn apart. In the end, all you have is molecules (or ions) of the solute floating around in the water. Here's another way of looking at it: |