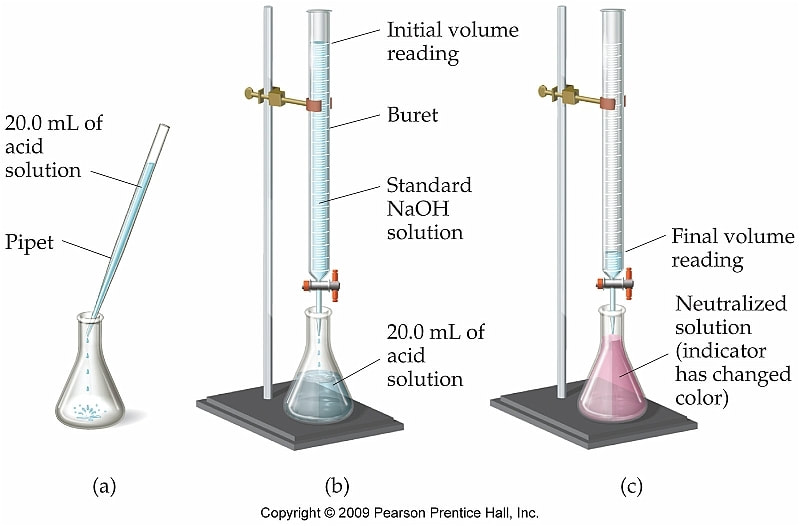
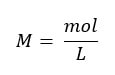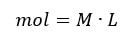Titration
|
Titration is a way to identify unknown concentrations of acids or bases.
In titration reactions, you neutralize an unknown acid with a known concentration of base. By knowing the amount of moles of base added, you can determine the moles and molarity of acid. Process: In a Erlenmeyer flask, you will place a certain amount of acid that you do not know the concentration of. Above the flask, in a buret, you would place a neutralizing base. Fill the buret to the zero mark with a base that you know what the concentration is. Then, drain enough base out of the buret to bring the pH to 7 (neutral). This can be accomplished using an indicator (a substance that changes colors when it is in an acid or base) or by using a pH meter (which can measure the pH). A picture of a titration is at right: The equation to determine the molarity of your unknown acid (or base) is: |
MaVa = MbVb
Where Ma stands for the molarity of the acid, Va stands for the volume of the acid, Mb stands for the molarity of the base and Vb stands for the volume of the base.
The reason why this works is because when you mix equal moles of acid and base, you get a neutral reaction. In the molarity equation:
so, if the moles are equal, we can set M L = M L and if the L's become V, then the equation above is what you get.
Example: 20 ml of an unknown concentration of acid is neutralized by 12.5 ml of 2.5 M base. What is the molarity of the acid?
Ma(0.020 L) = 2.5 M (0.0125 L)
Ma = 1.56 M
Here is a visual demo, and another example of the calculation: