Temperature
Gases can be measured in terms of:
|
Measurement
Amount Volume Pressure Temperature |
Standard Unit
mole liter atmosphere Kelvin |
We have already discussed amount, measured in moles, and volume. So, here we will focus on temperature.
Temperature
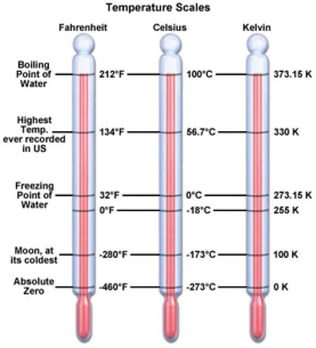
Temperature is the measure of the movement of the molecules in a substance. As heat energy is added to a material, the molecules of the material move faster (radiant heat energy transformed into kinetic energy of the molecules). Temperature is a measure of how fast the molecules are moving. Just like pressure, there is more than one temperature scales in use. In science, either the Celsius (OC) or the Kelvin (K) temperature scale is used. The conversions for these scales and the Fahrenheit (OF) scale are below:
|
oC = 5/9(oF - 32)
oF = 9/5(oC) + 32 K = oC + 273 oC = K - 273 Absolute Temperature
An absolute scale means that it has limits on one or both ends of the scale. The Kelvin temperature scale cannot go below 0 K because this temperature corresponds to the point where the motion of the particles (their kinetic energy) ceases. If there is no movement of the particles, there is no temperature. This point is called absolute zero. In other words, it is physically impossible to go below the following temperatures: 0 K = -273 oC = -459.4 oF
|