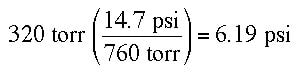Pressure
Gases can be measured in terms of:
|
Measurement
Amount Volume Pressure Temperature |
Standard Unit
mole liter atmosphere Kelvin |
|
We have already discussed amount, measured in moles, and volume. So, here we will focus on pressure.
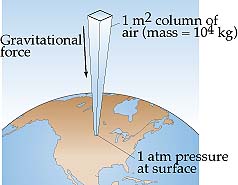
Pressure is the result of the gas particles colliding with the walls of the container it is in. Every time a particle hits the wall of the container, it exerts a force or push. Pressure is a measure of this force over the whole container. When the particles hit either with more force or more often (both due to faster molecule movement), the pressure will increase. Atmospheric pressure is the weight of the air above an object. When you are standing at sea level, there is a lot of air above you. All that air is essentially sitting on your shoulders, holding you down. That weight of gas at sea level is 14.7 pounds of pressure on every square inch of your body. You do not feel this weight because your body is used to it. It has been there every day of your life. Since 14.7 psi is a hard number to work with, scientists have developed an alternative unit as the standard. The atmosphere (atm) is equal to 14.7 psi. The unit was developed simply: At sea level, a person has exactly one Earth's atmosphere of air above them. Therefore, the air pressure at sea level is 1 atmosphere. As a person travels up from sea level, the amount of air above that person is less, or that person has less than one atmosphere above them. What would that atmospheric pressure be in Death Valley, which is 100 ft below sea level? Units of Pressure |
There are several other common units of pressure (just like there are several ways to measure distances). These units were all developed in different ways, but all of these are equal to one another at sea level
1 atmosphere = 760 mm Hg = 760 torr = 101,325 Pa = 101.325 kPa = 14.7 psi
Example of a pressure conversion: Suppose the pressure on a balloon in mid-air is 320 torr. What is the pressure in psi?