Chemical Bonding
Drawing Ionic Compounds
Using the Lewis structures, we can show the ionic bonding of compounds.
In ionic compounds, electron(s) transfer from one element to another, making cations (positive ions) and anions (negative ions) that attract one another.
Ex: Sodium Chloride (NaCl)
Using the Lewis structures, we can show the ionic bonding of compounds.
In ionic compounds, electron(s) transfer from one element to another, making cations (positive ions) and anions (negative ions) that attract one another.
Ex: Sodium Chloride (NaCl)
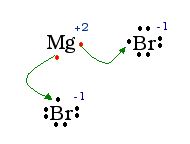
Explanation: Na needs to lose one electron to obtain an octet. Cl must gain one to get an octet. The valence electron from the Na is moved to the Cl. When this happens, Na achieves a +1 charge, and Cl become -1. The charges pull the two atoms together.
Another example: MgBr2
Drawing Covalent Compounds
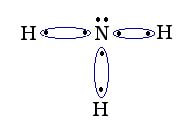
Another way that compounds can form is when the electrons are shared between 2 or more atoms to complete an octet. This results in a molecule with covalent bonds. A molecule that combines covalently is ammonia NH3 :
|
Explanation: N has 5 valence electrons. Each H has 1 valence electron. When H and N share electrons, both atoms can claim the electrons in the sharing. Therefore, H can claim it has two electrons. With the first bond, the valence electron count for N is 6. The N then bonds with the other two H and achieves an octet.
Very often in covalent compounds, there are unshared pairs of electrons which belong exclusively to one of the atoms. This is OK. It is not necessary to have bonds coming off of all the valence electrons. |
Multiple Bonds
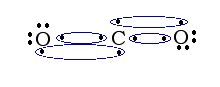
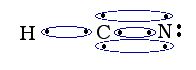
Two atoms may form double or triple covalent bonds in which multiple pairs of shared electrons are used to satisfy the octet rule.
Two atoms may form double or triple covalent bonds in which multiple pairs of shared electrons are used to satisfy the octet rule.