Naming Hydrocarbons
|
Alkanes, alkenes and alkynes are the basis of an organic substance. You start with one of these, and then can add polymers or functional groups as necessary. Hydrocarbons have a different naming system than other inorganic compounds.
Naming System for Organics Alkanes are a saturated hydrocarbon, with all single bonds. When naming an alkane, it is necessary to follow these two steps: 1) The prefix of an alkane is determined by the number of carbon atoms of the main chain. Prefixes are listed to the right: 2) Followed by an -ane ending. Example: CH3-CH2-CH3 is called propane, because it has 3 carbons, and is connected by single bonds.
|
Number of Carbons
1 2 3 4 5 6 7 8 9 10 |
Prefix meth- eth- prop- but- pent- hex- hept- oct- non- dec- |
Alkenes and Alkynes:
Alkenes and alkynes are unsaturated hydrocarbons. Alkenes have double bonds and alkynes have triple bonds. As long as a hydrocarbon has one double bond, that automatically makes it an alkene. The naming system has the same prefixes, but the name ends with -ene for alkenes and -yne for alkynes
Examples:
Alkenes and alkynes are unsaturated hydrocarbons. Alkenes have double bonds and alkynes have triple bonds. As long as a hydrocarbon has one double bond, that automatically makes it an alkene. The naming system has the same prefixes, but the name ends with -ene for alkenes and -yne for alkynes
Examples:
1) CH3-CH=CH-CH3 is called butene (4 carbons, double bond)
2) CH3-C≡C-CH3 is called butyne (4 carbons, triple bond)
2) CH3-C≡C-CH3 is called butyne (4 carbons, triple bond)
Numbering Addition
It is necessary to tell where the double and triple bonds are on the molecule. For this purpose, we list a number at the beginning of the name to tell where the double or triple bond is starts. When numbering, it is possible to number right to left, or left to right. The lowest number possible needs to be listed.
Examples:
It is necessary to tell where the double and triple bonds are on the molecule. For this purpose, we list a number at the beginning of the name to tell where the double or triple bond is starts. When numbering, it is possible to number right to left, or left to right. The lowest number possible needs to be listed.
Examples:
1) 2-pentene ---- CH3-CH2-CH=CH-CH3
2) 3-hexyne ---- CH3-CH2-C≡C-CH2-CH3
2) 3-hexyne ---- CH3-CH2-C≡C-CH2-CH3
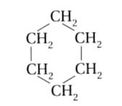
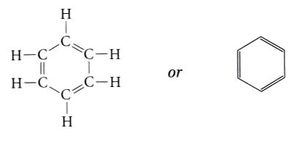
Any hydrocarbon molecule that is circular (3 carbon minimum) will have a cyclo- prefix at the beginning.
Examples:
Examples:
This video applies up through 14:30.