Energy Diagrams & Enthalpy
Energy Diagrams
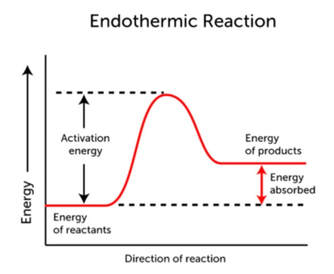
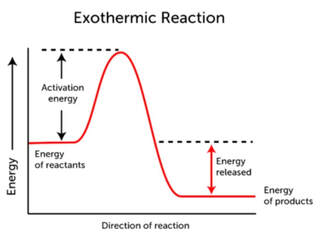
The heat of a reaction depends on the chemical energy of the reactants in comparison to the chemical energy of the products. Most reactions require some energy to get the reaction started. This energy is called the activation energy. The activation energy is the energy necessary to break apart all of the molecules into atoms. Sometimes, the substances have enough energy within them to achieve activation energy, and sometimes more energy is needed. Then, as the bonds reform, the higher amount of energy is used to reform bonds. Typical energy diagrams look like this:
The heat of a reaction depends on the chemical energy of the reactants in comparison to the chemical energy of the products. Most reactions require some energy to get the reaction started. This energy is called the activation energy. The activation energy is the energy necessary to break apart all of the molecules into atoms. Sometimes, the substances have enough energy within them to achieve activation energy, and sometimes more energy is needed. Then, as the bonds reform, the higher amount of energy is used to reform bonds. Typical energy diagrams look like this:
|
In the endothermic reaction on the left, energy is put into the reaction in order to break all the bonds. That is the activation energy that gets the total energy to the top of the energy "hill". The energy then goes down as bonds are formed. In the endothermic reaction, the energy required to make the products is more than the energy stored in the reactants to begin with. Since there is a net increase in energy (more energy has gone in than released), this is called an endothermic reaction.
|
|
The exothermic reaction to the right, still has an activation energy, but after the top of the energy "hill" is achieved, the "fall" in energy to the products ends up below the energy of the reactants. Since, overall, the reactants had more energy in them than the products, the extra energy not used goes out from the reaction. This net decrease in energy (more energy has been lost than gone in) make this reaction exothermic.
|
Enthalpy
Enthalpy is a measure of the heat energy absorbed and released in a reaction. Enthalpy is figured as the change in the amount of energy as the reaction continues. The symbol for enthalpy is ΔH. It is based upon the internal potential energy of the reactants (Hreactants) and the internal potential energy of the reactants (Hproducts).
Enthalpy -> ΔH = Hproducts - Hreactants
The enthalpy is either included in the equation:
2 C2H6 + 7 O2 --> 4 CO2 + 6 H2O + 3120 kJ
Or at the end of the reaction:
2 C2H6 + 7 O2 --> 4 CO2 + 6 H2O, ΔH = -3120 kJ
In the case of the above reaction, the reactants contain more potential energy than the products. Therefore the ΔH is negative. A negative enthalpy means the reaction is exothermic because the reactants have lost energy while making the products.
Here are examples of an endothermic reaction. Notice that the when the enthalpy is in the equation, it is on the reactant side, indicating the energy needed to make the reaction occur:
2 SO3 + 196 kJ --> 2 SO2 + O2
At the end of the reaction, the enthalpy is positive, to indicate energy added to the reactants:
2 SO3 --> 2 SO2 + O2, ΔH = 196 kJ
Quiz yourself (answers at bottom):
Endothermic or exothermic?
1) Heat is added to the reaction to make it occur.
2) - ΔH
3) kJ unit on the right side of the equation
4) Heat energy is a product
5) + ΔH
6) kJ unit on the reactants side
Endothermic or exothermic?
1) Heat is added to the reaction to make it occur.
2) - ΔH
3) kJ unit on the right side of the equation
4) Heat energy is a product
5) + ΔH
6) kJ unit on the reactants side
Answers to quiz:
1) endo
2) exo
3) exo
4) exo
5) endo
6) endo
1) endo
2) exo
3) exo
4) exo
5) endo
6) endo