Water Softening
Hard Water
Hard water is water containing an excess of calcium (Ca+2), magnesium (Mg+2) and iron (Fe+3) ions. Many parts of the country have hard water, as shown by the following diagram:
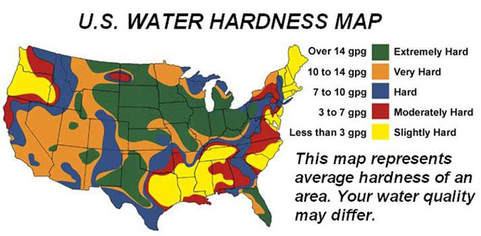
These ions, when present in water, will combine with soap to form insoluble compounds. These insoluble compounds do not rinse away easily, and can leave soap deposits on dishes and on your skin. This is not really harmful, but there are incoveniences: Hard water ions will cause fading of colors on clothing, will cause a white film to appear on dishes and can dry out your skin. The hard water can also cause calcium carbonate deposits to form in pipes and in water heaters, which reduces efficiency and flow through the pipes.
To remedy the inconveniences of hard water, many households where hard water is present will have a water softener, which transforms hard water into soft water.
To remedy the inconveniences of hard water, many households where hard water is present will have a water softener, which transforms hard water into soft water.
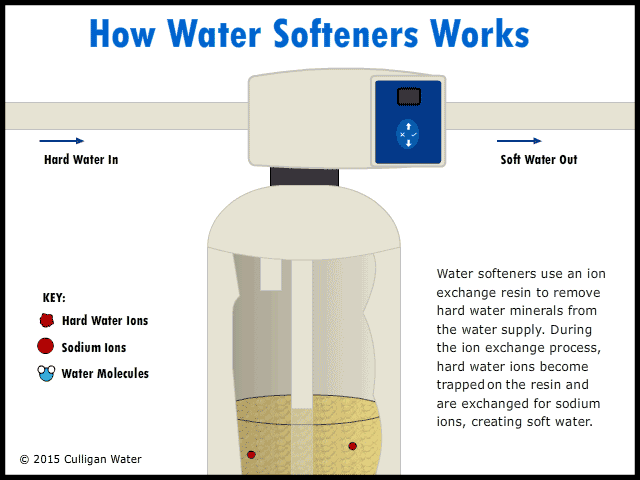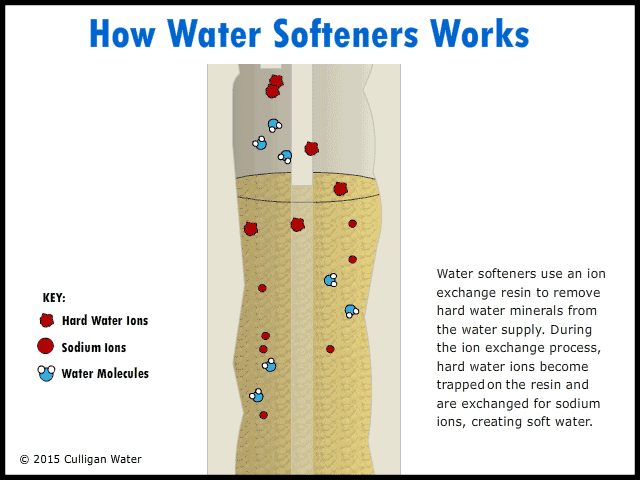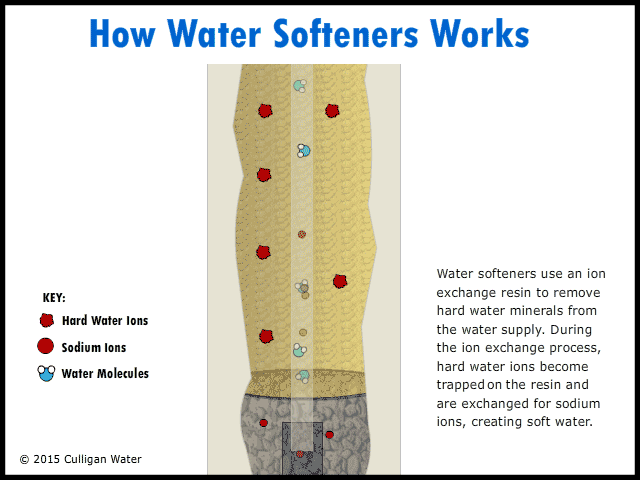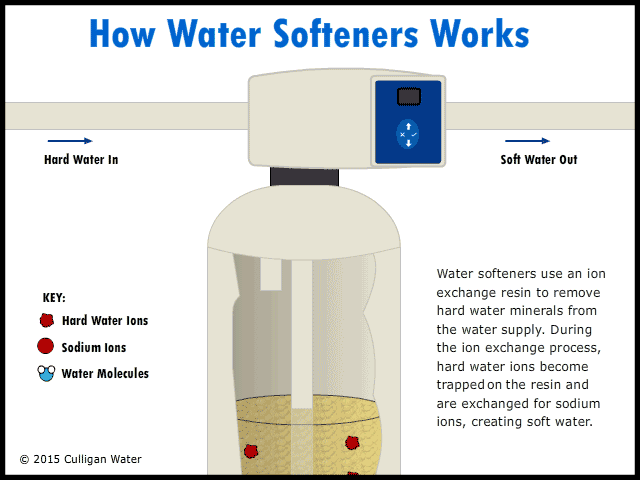
Operation of a Water Softener
|
The water enters your house, and the pipe goes directly to the water softener. As the hard water flows into the water softener, the hard water comes in contact with an ion exchange resin. The resin absorbs the hard water ions of calcium, magnesium and iron. As the hard water ions are absorbed, ions of sodium are released into the water. This soft water ions does not affect the cleaning abilities of soap, and is harmless. As the water runs, the ions are replaced throughout the day. The soft water goes up through the center of the softener and then into your house faucets.
At night, when water is not being used, the valves on the left in the figure above close, closing off the water coming into the house. Salt water is then pumped into the water softener. The salt water flows into the ion exchange resin, and the sodium in the salt replaces the hard water ions that have been absorbed into the resin. The resin becomes saturated with sodium ions, and the hard water ions are rinsed out of the water softener to the waste. This process regenerates the ion exchange resin. The recharged resin can the soften more water on the next use. |